Ali Phaneuf is a junior at Fairfield University and was a page in the Reader’s Services Department this past summer. As a journalism major and a studio art minor, Ali has always been an avid book reader and art enthusiast, and her love of books and creativity was able to grow through her experience at AAS.
As a Studio Art minor, I’m fortunate enough to be constantly exposed to new and unique art courses at Fairfield University. A class I took in the spring of my sophomore year proved to be one of my most challenging thus far, as I was thrust into a time machine that took me back to the art of nineteenth-century photography—well, for the most part. Throughout the semester I had the opportunity to create prints in nineteenth-century styles, including cyanotype, kallitype, and albumen; however, we simulated these nineteenth-century processes using digital technology.
When I returned to AAS after completing my sophomore year at Fairfield, I showed some of my prints to fellow co-workers in the Readers’ Services Department, who then directed me to photo albums of cyanotype prints in the AAS collections. Thanks to the help of Ashley Cataldo, assistant curator of manuscripts, and Lauren Hewes, curator of graphic arts, I was able to view the album of Lyford J. Chauncey and photos contributed by the Worcester Natural History Society. The AAS photo album of Lyford J. Chauncey includes 101 cyanotypes. Each cyanotype has a caption written underneath the mount, and most of the images are landscape views of locations in Spencer and Worcester, Massachusetts. These beautiful images inspired me to create some of my own cyanotypes as they would have been created during the nineteenth century.

Another book at AAS that was particularly interesting to me included the work of Anna Atkins, one of the first cyanotype artists. Rather than using cyanotype to develop a negative, Atkins used it to showcase botany through photographs. She would take flowers and plants and place them on the paper coated with the cyanotype chemicals. She then placed the contact frame outside in the sunlight, just as photographers would when developing a negative. After the print had reached its prime exposure time, Atkins would remove the plants from the frame and rinse the cyanotype under cold water to develop an image showing the shape of the flower or plant. Atkins’s creativity and ingenuity contributed to my enthusiasm for the cyanotype process.
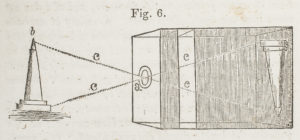
Many people are familiar with the term “camera obscura,” which literally means “dark room”, as explained by A History and Handbook of Photography by Gaston Tissandier, held at AAS. The concept of the camera obscura was first used in the later part of the sixteenth century by philosophers who discovered that making a small opening in the shutter of a window—which was so tightly closed no other light could get through—allowed the rays of light to penetrate through the small hole and project onto a white screen a reverse image of exterior scenery or objects.

Early photographers would use the camera obscura to then trace the image reflected into the room to create a dimensionally accurate illustration of the outdoors. Camera obscuras were later used in a portable manner. Eventually, light-sensitive material adhered to glass or fiber-based paper were used within the camera obscura to retain an image.
I created my own camera obscura by painting a shoe box completely black. I then used an X-acto knife to cut out a small square on one side of the shoe box, which I then filled with aluminum foil. To make my small hole in the camera, I poked a minute opening through the foil with a thumb tack. I had my own nineteenth-century portable camera obscura, also known as a pinhole camera.

Before I could work with the light-sensitive paper, I had to somehow create a low-cost dark room. The place in my home with the fewest windows and the least amount of light is my dad’s basement workroom. There is only one small rectangular window, which I covered with a black trash bag to make the room completely dark. I unpacked the desk lamp I use at college and put a red light bulb in it so that I could see what I was doing while in my makeshift darkroom. I was finally ready to get started.
For light-sensitive photo paper, I used Ilford Multigrade black and white glossy paper and taped a piece to the inside of the shoebox opposite the hole. I then closed the box and covered the hole with a piece of black electrical tape so that light would not shine into the box prematurely. I went outside on a clear sunny day and set up the camera in my backyard. I removed the tape for a little less than one minute, placed it back on, and then went downstairs into my dark room. In order for the image to appear and remain permanent when in sunlight, I had to remove the paper from the camera and insert it into a tray filled with liquid developer composed of water, sodium carbonate, sodium sulfite, hydroquinone, and (methylamino) phenol sulfate. I then continued to put the print through a second tub of water, followed by a water, sodium thiosulfate, sodium metabisulphite, boric anhydride, and sodium tetraborate fixer. I then finished by rinsing the print off with more water. The results from my first attempt using a self-made camera obscura were less than underwhelming—a solid black image is what appeared.

This could mean one of two things: I exposed the image for too long, or my camera obscura was not sealed tightly enough and extra light was coming in through the sides. I decided to focus my energy on the latter issue. For my second attempt, I sealed my box tighter, wrapping all sides with black electrical tape. However, the results were yet again a solid black image. I then decided to shift my focus towards timing. I cut my exposure time in half and left the pinhole open for about thirty seconds. This time I finally started to see some sort of dull image. Then, I tried shooting my images from the shade, rather than from the direct sunlight. After opening the pinhole for about thirty seconds in the shade, I finally received a clear Image of my backyard — yes, this means I actually took a picture using a shoebox!
So, how does it work? Each point of the scene that the camera is angled towards emits light, which passes through the pinhole and creates a point of light on the back of box (onto the light-sensitive paper). The light-sensitive paper records the image that is projected onto the back of the box, which is how the picture is created.
When light shining through the pinhole hits the Ilford photo paper, it causes a chemical reaction with the silver-halide crystals in the paper. This reaction causes the crystals to turn black when processed with developing chemicals, thus the parts hit with the most light (the brightest parts of the scene) turn the blackest. This inverted image is referred to as a negative. In order to make this negative image into a positive image, photographers would take the negative and develop it using an alternative process. I chose to use the cyanotype process.
The cyanotype process was first invented by Sir John Hershel in 1842 and requires the use of natural fiber paper (I used watercolor paper). Hershel’s formula requires one mixture of 20g of ammonium citrate with 100ml of water and a second mixture made up of 16g of potassium ferricyanide with 100ml of water. The process then requires the artist to mix these two formulas together in a 1:1 ratio in an amount that’s only required to saturate the paper (for example, I used about 12 drops of each solution to make a small mixture). I then poured the solution that I mixed onto the watercolor paper and brushed it over the paper using a foam brush (the combination of the two formulas should result in a greenish color on the paper). I let the paper dry overnight, and the next day I was ready to print my image. I placed the negative image face down on top of the paper coated with the cyanotype mixture and put the two papers into a glass picture frame to press the two papers together, which I then laid outside in direct sunlight like so:

When placing the negative image on top of the cyanotype, values are reversed to their original state as they appeared in “real life.” When light shines through the negative, the dark parts block the most light. This blocking of the sun results in those spots remaining the greenish color, whereas the rest of the image should appear dark silver. Once you see this silver color appear on your print, you can take the print inside to be developed. Unlike developing the negative, developing the cyanotype is a lot less complicated. I simply went back into my darkroom and put the print into a bin of cold water. The greenish color washed away (making those areas white) and the silver colors became blue. Once rinsed in water, the print is safe to bring back into the sunlight. As the print is exposed to light longer, the chemistry becomes darker and the tonal ranges intensify. To gain a greater Prussian blue, cyan color, I inserted the print into a wash of lemon juice to achieve my final product.
Lyford J. Chauncey’s photos were printed during the years 1885 to 1900, meaning that Chauncey may have used a camera that didn’t differ too much from my shoebox. Chauncey most likely used some sort of box camera composed of wood and metals. There were many different types of box cameras that emerged after 1880, including the Change-Box Camera, Magazine Camera, and Reflex Camera. The key difference would have been that Chauncey used a lens with his camera rather than a simple hole, which would have resulted in clearer images. It is also possible that Chauncey used a glass negative or even film, rather than the photo paper I used. However, after looking through the album, Chauncey’s work demonstrates some of the same struggles that I faced. Some of his images appear very dark—a result from a large amount of chemicals being used to saturate the fabric paper or overexposure in the sunlight—while other images appeared very faint—a result of not enough chemical solution or not enough exposure to the sunlight. Below are some Chauncey images that inspired my own cyanotypes.



I even made an attempt at a nineteenth-century “selfie.” I had to set up my shoebox on a milk crate in order for the pinhole to be the right height and have my face in view. Then, I quickly removed the black electrical tape from the pinhole, ran into place, held my pose for about one minute, and then ran back to the camera to re-cover the hole. I appear a bit blurred in the image because of my racing to uncover and recover the hole in the camera.
Throughout the summer I learned that photography is a complicated process that comes in many forms, styles, and appearances. From using a small portable box to not using a camera at all, photographs can be made with almost anything. Chauncey used a photographic process to capture the beauty of his surroundings, while Atkins worked to preserve the natural form of botany through this same process. How a photographer or artist hopes to preserve certain scenes, objects, or memories is an individual decision, which can allow their own thoughts to radiate through their work. I’ve learned that many factors went into nineteenth-century photography. Such elements as the chemical mixture, sunlight density, and exposure time all made these alternative processes a somewhat tedious, yet fascinating art form. This summer I was lucky enough to discover the beauty in practicing something old to create something new with the help of something blue.

